IVDR transfer process
-
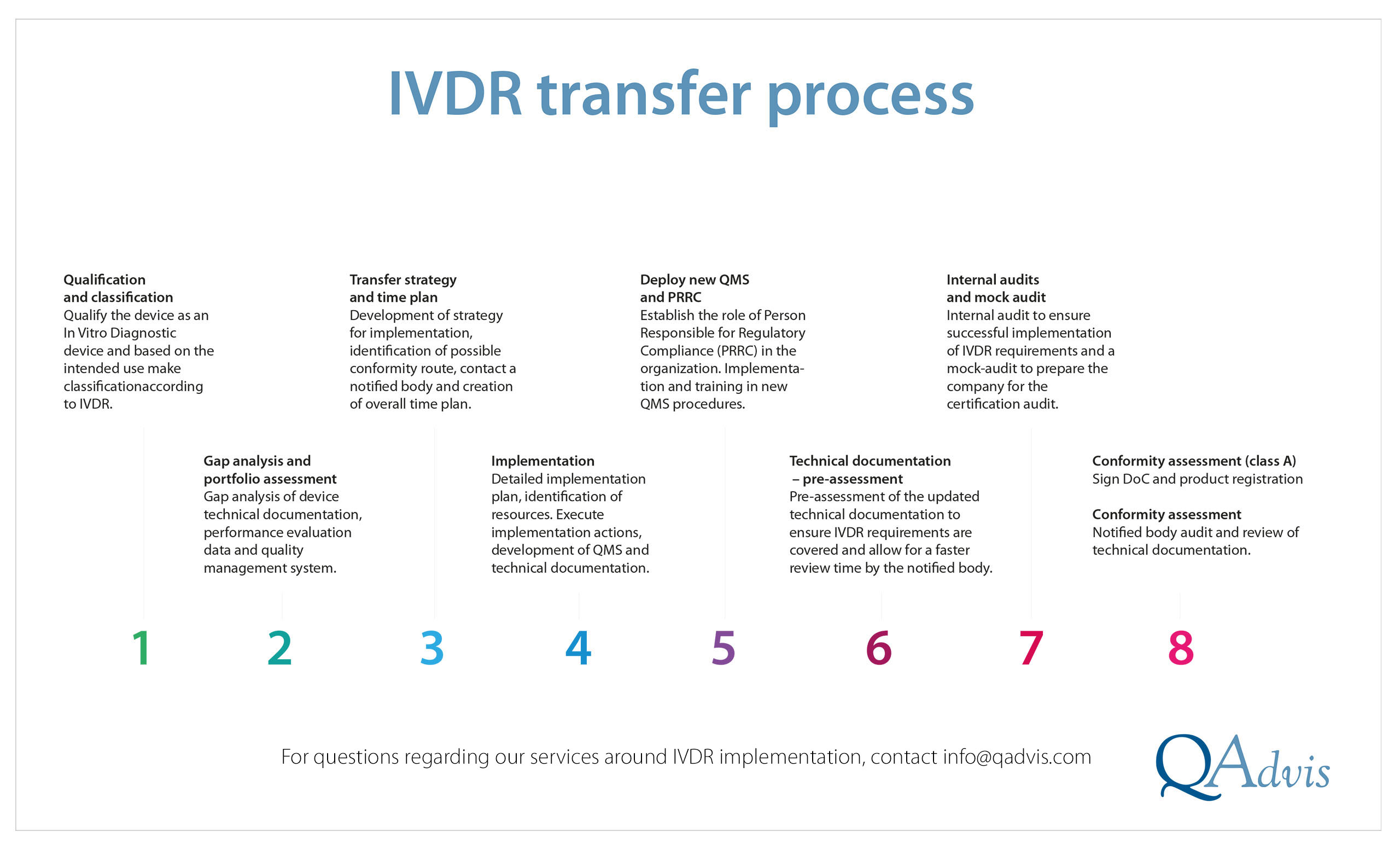
Qualification and classificationStart with qualifying the device as an in vitro diagnostic medical device and based on the intended purpose classify the device according to the classification rules in IVDR.QAdvis consultants can support you with review of intended purpose, together with qualification and classification rational for your devices.
-
Gap analysis and portfolio assessmentGap analysis of device technical documentation, clinical data, and quality management system, where QAdvis can support with expertise in the areas needed.
-
Transfer strategy and time planDevelopment of strategy for implementation, identification of possible conformity route, contact a notified body and creation of overall time plan for the implementation project. We can help you develop the project plan and if needed also provide support in notified body identification and contacts.
-
ImplementationDetailed implementation plan and identification of resources. Execute implementation actions, development of QMS and technical documentation.
Implementation action plan as a complement to the project plan might be needed. Updated your technical documentation and QMS according to the project plan, we are aware that this can be time consuming and require resources and expertise within several areas and our consultants are available to fill the gaps.
- Deploy new QMS and PRRC
As a manufacturer of medical devices, there is a need to establish the role of Person Responsible for Regulatory Compliance (PRRC) in the organization. As well as implementation and training in new and updated QMS procedures for all employees.QAdvis have regular IVDR and QMS training sessions, as well as on-site training on request. -
Technical documentation – pre-assessmentReview of technical documentation by the notified body ca be time consuming and expensive, planning for a pre-assessment of the updated technical documentation to ensure IVDR requirements are covered and allow for a faster review time by the notified body.Our experts in technical documentation can make this review to help prepare and avoid common pitfalls. Delivering a review report with identified proposed actions for the technical documentation.
-
Internal audits and mock auditIVDR requirements need to be included in the internal audit program, and an initial audit is recommended to ensure successful implementation of IVDR requirements, as well as a mock-audit to prepare the organization for a successful certification audit.QAdvis auditors with vast experience from medical device audits, can perform an audit and identify proposed actions and non-conformities to be corrected before the notified body audit.
-
Conformity assessmentAs a final step, the conformity assessment is conducted, QMS audit and review of technical documentation by the notified body.QAdvis can support you during the audit as well as with handling eventual non-conformities after the audit.
Contact me for further information
Per Sundström
Office: +46 8 621 01 05 or send me an email